- Blog
- Fabfilter pro q 2 vst windows kickass
- Game maker studio 2 cracked
- Ab system mestrenova
- Siemens simatic s7-1200 models
- Jonathan anderson neurotracker
- Ley lines through michigan
- Server os market share
- Spotflux indir
- Dior addict gloss 853 rouge defendu
- Eeprom programmer download
- Chiff and fipple how to make keys flute
- Autocad 2009 30 day trial
- Best video capture device for vhs transfer
- Corpse party blood drive english full
- Bloons td battles pc shortcuts
- Age of empires iv microsoft
- Where to download ptedit32
- Mac folder icon png
- Copy dos2usb license to new pc
- Isilo app for ipad
- Jayam telugu movie songs to download
- Navionics web
- Maui revealed book pdf
- Oh meri madhubala song
- When to put a dog down with torn acl
- Naked and funny gym
The kaolinite structure is one of the kaolin sub-group polytypes and is made of dioctahedral 1:1 layers with the Al 2Si 2O 5(OH) 4 general composition ( Brigatti et al., 2013). Kaolinite has potential uses in more specialized and advanced application niches if the interlayer chemistry that results from grafting or intercalating organic moieties can be better understood, mastered, and developed ( Dedzo and Detellier, 2016). In that context, well developed characterization methods are needed to precisely monitor kaolinite structural modifications that are related to intercalation. Hence, it is important to understand the kaolinite structural modifications and surface functionalization used to control a vast spectrum of applications and processes, such as catalysis and organic reactivity (Laszlo, 1986 Mccabe and Adams, 2013, Ngnie et al., 2016), clay-polymer nanocomposites ( Letaief and Detellier, 2009 Galimberti et al., 2013), bitumen extraction from oil sands ( Detellier et al., 2015 Lin et al., 2016), soil permeability and soil transport processes ( Yong and Mourato, 1990), or the abiotic origin of life ( Brack, 2013). Kaolinite plays a prominent role in human activities ( Schroeder and Erickson, 2014). paper coating, paint, ceramics, etc.) ( Murray and Keller, 1993 Murray, 2007). Kaolinite is very abundant in nature and large quantities are used in classical applications (e.g. Both the 1H and 27Al MAS NMR studies at different magnetic fields afforded important information about the local environments of the kaolinite hydroxyl groups and structural Al(III).Ĭlay minerals are known for many unique chemical and physical properties that make them attractive starting materials for a vast variety of applications ( Johnston, 2010 Bergaya and Lagaly, 2013). Increased quadrupolar constants were observed and showed the major perturbations of the local Al symmetry that resulted from DMSO intercalation. The two octahedral Al(III) sites are not equivalent and can be distinguished in the low-field spectral simulation. The 27Al MAS NMR spectra of KGa-1b obtained under different magnetic fields revealed that most of the quadrupolar effects were highly reduced at 21.1 T, whereas the spectra at lower field, 4.7 T, were dominated by quadrupolar effects. The 1H MAS NMR chemical shifts of the two methyl groups in DMSO-K are not equivalent and can be attributed to the 2.9 and 4.2 ppm peaks. In the present work, the 1H MAS NMR chemical shifts of KGa-1b were unambiguously attributed to the internal surface hydroxyls at 2.7 ppm and to the internal hydroxyls at 1.7 ppm. Because quadrupolar interactions are sensitive to the local octahedral Al(III) geometry, solid-state 27Al NMR can follow subtle structural modifications in the octahedral sheet. 1H NMR, nevertheless, can give unique information about kaolinite hydroxyls. The use of 1H and 27Al MAS NMR for this purpose has been relatively rare. Well developed characterization methods are essential to define the structural modifications of kaolinite, and MAS NMR is a useful complement to other techniques. Intercalating DMSO into kaolinite to form the DMSO-K intercalate is, thus, a particularly useful first step toward the intercalation of a large variety of molecules, including polymers and ionic liquids. Once the interlayer space is expanded, the intercalated compounds can be replaced in a second step.
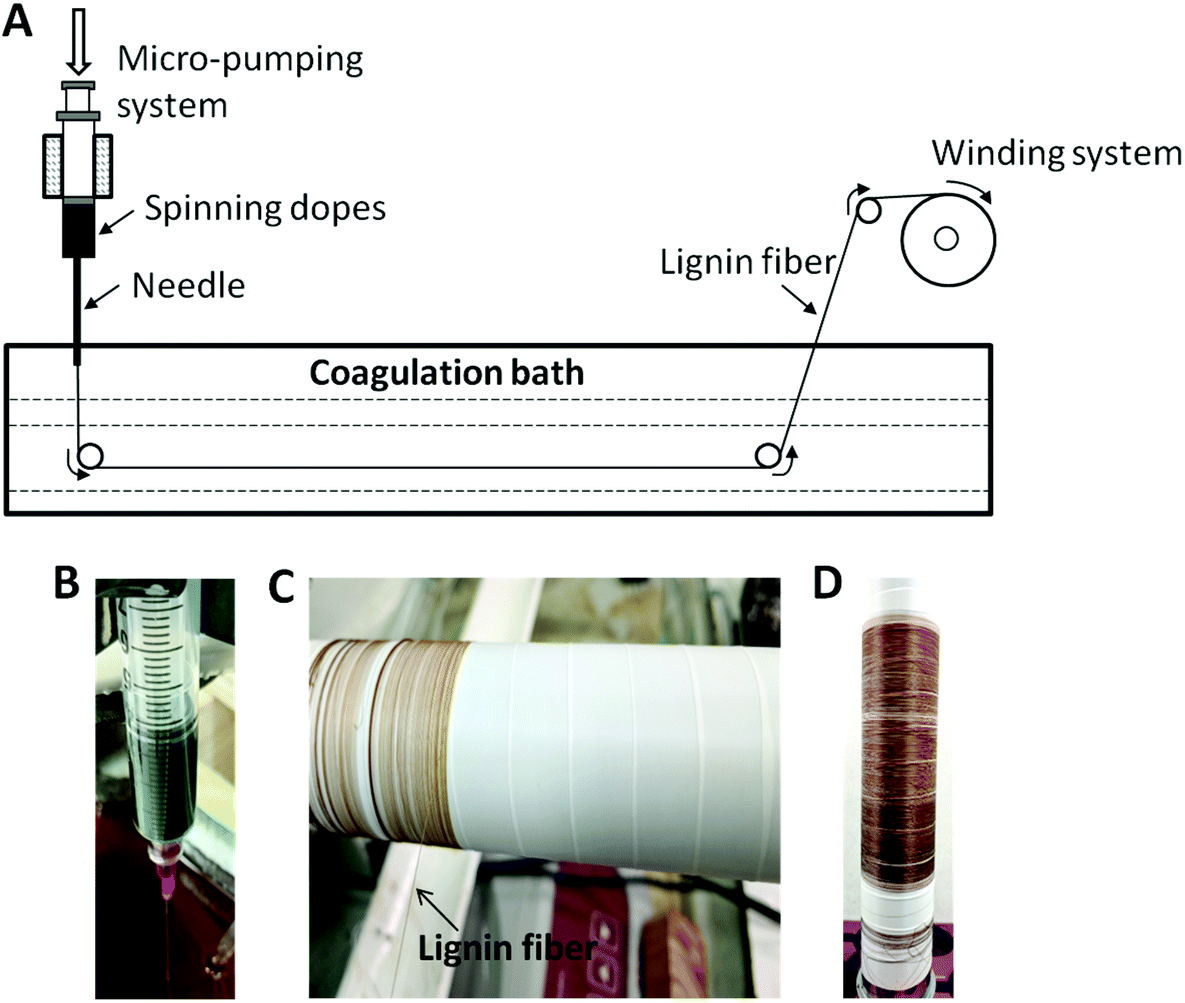
The interlayer chemistry of kaolinite (K) was examined by intercalating a select group of highly polar organic molecules or salts into kaolinite as a first step.

In this work, unique structural information on kaolinite and on kaolinite dimethylsulfoxide (DMSO) intercalate were provided by solid-state 1H and 27Al magic-angle spinning (MAS) NMR.
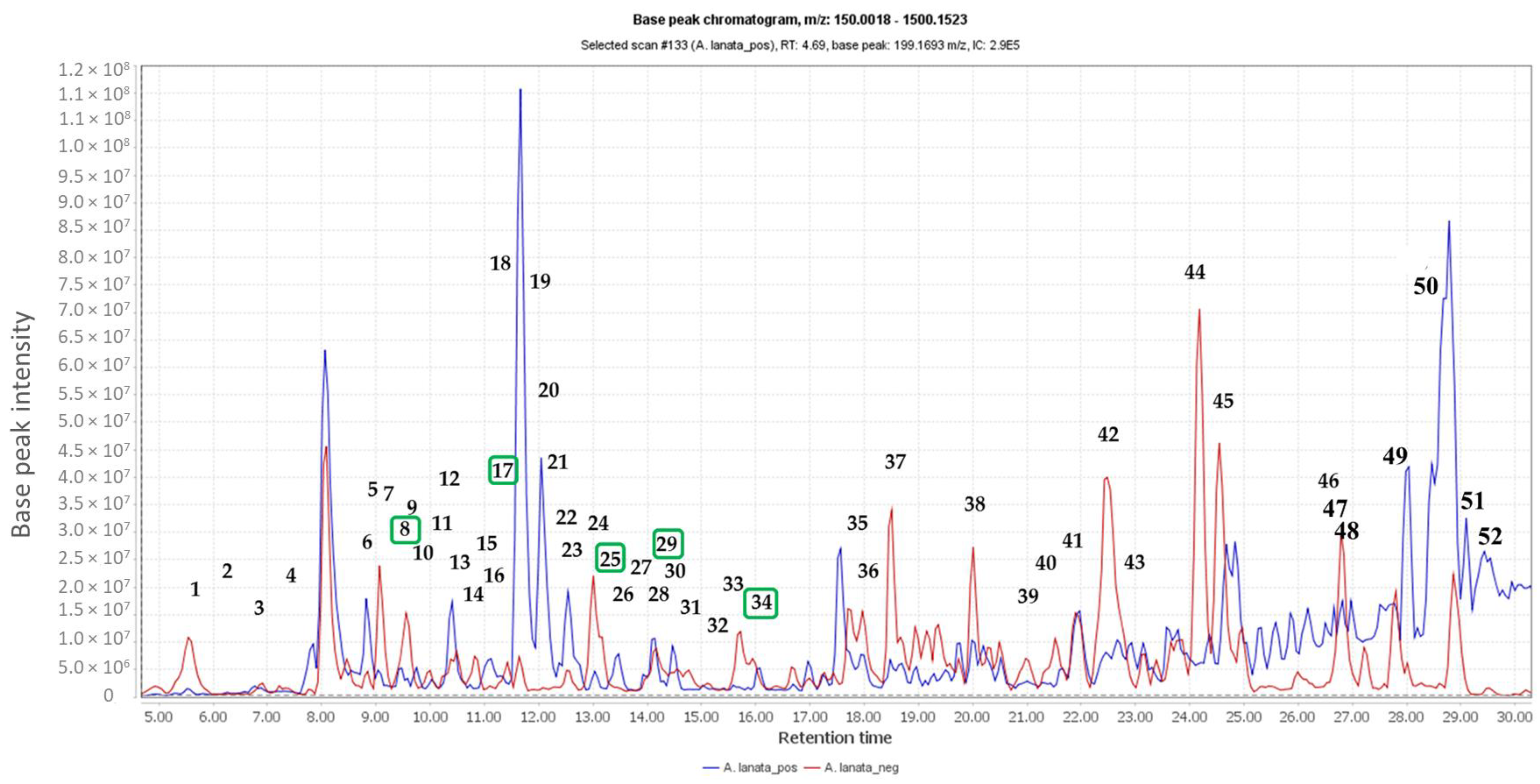
The new diorganodiselenides + − and + − were established by single-crystal X-ray diffraction.Nuclear magnetic resonance (NMR) provides a powerful tool to describe local nuclear environments.
- Blog
- Fabfilter pro q 2 vst windows kickass
- Game maker studio 2 cracked
- Ab system mestrenova
- Siemens simatic s7-1200 models
- Jonathan anderson neurotracker
- Ley lines through michigan
- Server os market share
- Spotflux indir
- Dior addict gloss 853 rouge defendu
- Eeprom programmer download
- Chiff and fipple how to make keys flute
- Autocad 2009 30 day trial
- Best video capture device for vhs transfer
- Corpse party blood drive english full
- Bloons td battles pc shortcuts
- Age of empires iv microsoft
- Where to download ptedit32
- Mac folder icon png
- Copy dos2usb license to new pc
- Isilo app for ipad
- Jayam telugu movie songs to download
- Navionics web
- Maui revealed book pdf
- Oh meri madhubala song
- When to put a dog down with torn acl
- Naked and funny gym
